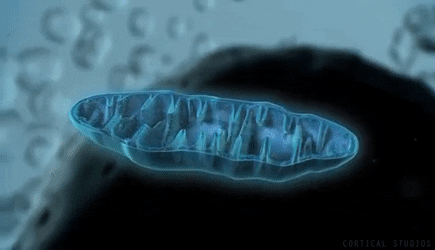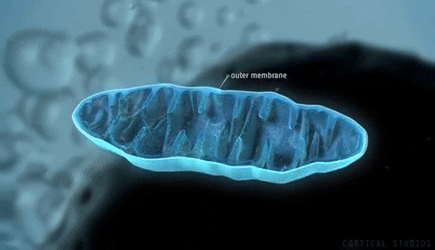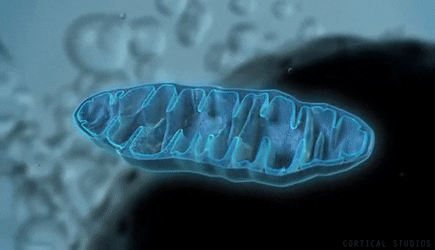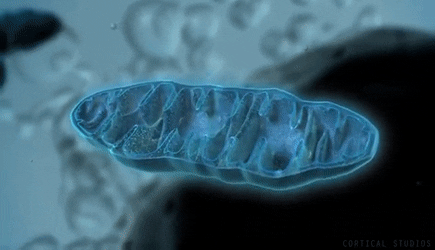
Of all the organelles to be found inside eukaryotic cells, the DNA-sheltering nuclei might be the best-known, but the mitochondria are surely not far behind. Mitochondria are familiar as bean-shaped structures floating in the cytoplasm, and they are almost inevitably referred to as “powerhouses” of the cell because they generate adenosine triphosphate (ATP), the fuel for most metabolic processes. For more than a century, biologists believed that energy production was their only role.
But that simple picture of mitochondria is turning out to be shockingly incomplete.
Mitochondria may look static and uniform in textbooks, but as researchers recognized early on, in reality the organelles change shape constantly through cycles of fusion (in which they combine and elongate) and fission (in which they split and shrink). They form highly dynamic, short-lived tubular networks threading throughout a cell. Recently, it has become clear that mitochondria also perform signaling and regulatory functions that are only indirectly related to their job as energy providers. In the past few years, research has revealed that one of their key roles is in controlling the development and ultimate role of stem cells.
Now scientists at the University of Ottawa in Canada have provided evidence that the morphing shapes of mitochondria powerfully influence neurogenesis, the development of neurons. In making this discovery, the scientists have pieced together a connection between the organelle’s shape transitions and how it carries out its signaling functions.
In 2016, Slack, Khacho and their colleagues reported the first evidence that mitochondrial shape-shifting is a key regulator of neural stem cell fate, the decision to self-renew or differentiate. By deleting genes that encoded key proteins for the fusion and fission machinery in mice, they discovered that a deficiency in fusion proteins reduced neural stem cells’ capacity to replenish themselves and encouraged the cells to become neurons. A loss of fission proteins, on the other hand, stimulated the stem cells to self-renew.
Their work showed that changes in the shape and architecture of mitochondria are among the earliest, most “upstream” signals to determine which way neural stem cells will go.
Given the previously established link between alterations in the fission and fusion machinery and neurodegenerative disorders, the team also investigated whether disrupting mitochondrial dynamics could alter the production of new neurons. When they knocked out fusion proteins in the brains of fully grown mice, they discovered that this disruption of the shape-shifting process reduced the number of new neurons produced in the animals’ brains and led to impairments in memory and learning.
Genetic defects are also known to alter mitochondrial fission and fusion in humans, but the idea that they might particularly influence stem cells hasn’t really been explored yet, Slack said. “What we’re working on now is trying to find new ways, through dietary or pharmacological means, to improve mitochondrial function in stem cells so we can maintain optimum learning and memory for as long as we can.”
Source:
https://www.quantamagazine.org/shape-shifting-mitochondria-direct-stem-cells-fate-20190318/
Leave a comment